Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Asami, Makoto; Watahiki, Hiromi; Oi, Takao; Makino, Hitoshi; Shibata, Atsuhiro; Kameo, Yutaka; Meguro, Yoshihiro; Ashida, Takashi
JAEA-Data/Code 2014-016, 37 Pages, 2014/09
A database on the analyses of samples obtained from contaminated water in the circulating system of Fukushima Daiichi Nuclear Power Station was built. This database contains the analyses of 25 samples of JAEA and 313 samples of TEPCO which have been published in FY 2011 to FY 2013. Also, as well as the analyses on contaminated water, the information on the stored and treated amount in accumulated water and the amount of produced waste, which has been published by TEPCO and which might be required in order to estimate the inventory of secondary waste (sludge, used vessels) generated by treatment of contaminated water are contained in this database. This technical report shows the function of this database and user manual with example and presents the FY2013 edition of database by appendix CD.
 TiO
TiO under varied surface condition
under varied surface conditionOlivares, R.*; Oda, Takuji*; Oya, Yasuhisa*; Tanaka, Satoru*; Tsuchiya, Kunihiko
Fusion Engineering and Design, 75-79, p.765 - 768, 2005/11
Times Cited Count:9 Percentile:53.19(Nuclear Science & Technology)no abstracts in English
Kimura, Takaumi; Kirishima, Akira*; Arisaka, Makoto
Kidorui, (47), p.43 - 56, 2005/11
no abstracts in English
 TiO
TiO
Ryan, O.*; Oda, Takuji*; Oya, Yasuhisa*; Tanaka, Satoru*; Tsuchiya, Kunihiko
JAERI-Conf 2004-012, p.136 - 139, 2004/07
no abstracts in English
Kirishima, Akira; Kimura, Takaumi; Tochiyama, Osamu*; Yoshida, Zenko
Journal of Alloys and Compounds, 374(1-2), p.277 - 282, 2004/07
Times Cited Count:27 Percentile:75.6(Chemistry, Physical)no abstracts in English
 -1,2-dichloroethylene decomposition in low-humidity air under electron beam irradiation
-1,2-dichloroethylene decomposition in low-humidity air under electron beam irradiationSun, Y.*; Hakoda, Teruyuki; Chmielewski, A. G.*; Hashimoto, Shoji*
Radiation Physics and Chemistry, 68(5), p.843 - 850, 2003/12
Times Cited Count:8 Percentile:49.89(Chemistry, Physical)no abstracts in English
Hakoda, Teruyuki; Kim, H.*; Okuyama, Kikuo*; Kojima, Takuji
Journal of Aerosol Science, 34(8), p.977 - 991, 2003/08
Times Cited Count:15 Percentile:54.94(Engineering, Chemical)no abstracts in English
Okamoto, Yasuharu*; Mochizuki, Yuji*; Tsushima, Satoru*
Chemical Physics Letters, 373(1-2), p.213 - 217, 2003/05
Times Cited Count:8 Percentile:24.96(Chemistry, Physical)no abstracts in English
Kimura, Takaumi; Nagaishi, Ryuji; Ozaki, Takuo; Arisaka, Makoto*; Yoshida, Zenko
Journal of Nuclear Science and Technology, 39(Suppl.3), p.233 - 239, 2002/11
no abstracts in English
Hakoda, Teruyuki; Hashimoto, Shoji; Kojima, Takuji
Bulletin of the Chemical Society of Japan, 75(10), p.2177 - 2183, 2002/10
Times Cited Count:8 Percentile:37.18(Chemistry, Multidisciplinary)no abstracts in English
Suzuki, Satoru; Sato, Haruo; Ishidera, Takamitsu; Fujii, Naoki*; Kawamura, Katsuyuki*
JNC TN8400 2001-031, 44 Pages, 2002/05
In order to quantify effect of temperature on diffusivity of deuterated water (HDO) in compacted sodium-bentonite, through-diffusion experiments were conducted at elevated tempemture from 298 to 333 K. Kunipia F (Na-montmorillonite content  98 wt. %; Kunimine Industly Co.) was compacted to a dry density of 0.9 and l.35 Mg/m
98 wt. %; Kunimine Industly Co.) was compacted to a dry density of 0.9 and l.35 Mg/m . Since smectite flakes were perpendicularly oriented to a direction of compaction, anisotropy of diffusivity was investigated parallel and normal to the preferred orientation of smectite. Effective diffusion coeficient D
. Since smectite flakes were perpendicularly oriented to a direction of compaction, anisotropy of diffusivity was investigated parallel and normal to the preferred orientation of smectite. Effective diffusion coeficient D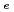 of HDO was larger for a diffusional direction parallel to the preferred orientation than normal to that for both dry densities. These results well agreed to the previously reported ones for tritiated water. Activation energies of D
of HDO was larger for a diffusional direction parallel to the preferred orientation than normal to that for both dry densities. These results well agreed to the previously reported ones for tritiated water. Activation energies of D in compacted bentonite increased with increasing dry density in the range of 19 - 25 kJ/mol which was slightly larger than that in bulk water (18 kJ/mol). This relationship can be considered to be due to both the pore structure development and high activation energy of water (18-23 kJ/mol) in the vicinity of smectite surface (within 2 nm) on the basis of molecular dynamics simulations.
in compacted bentonite increased with increasing dry density in the range of 19 - 25 kJ/mol which was slightly larger than that in bulk water (18 kJ/mol). This relationship can be considered to be due to both the pore structure development and high activation energy of water (18-23 kJ/mol) in the vicinity of smectite surface (within 2 nm) on the basis of molecular dynamics simulations.
Sugo, Yumi; Sasaki, Yuji; Tachimori, Shoichi
Radiochimica Acta, 90(3), p.161 - 165, 2002/04
Times Cited Count:153 Percentile:99.24(Chemistry, Inorganic & Nuclear)no abstracts in English
Watanabe, Shoichi; Miyoshi, Yoshinori; Yamane, Yuichi
JAERI-Tech 2002-043, 93 Pages, 2002/03
no abstracts in English
Ito, Akira; Kawakami, Susumu; Yui, Mikazu
JNC TN8400 2001-028, 38 Pages, 2002/01
In a repository for high-level radioactive waste, coupled thermo -hydro -mechanical and chemical (THMC) processes will ocurr, involving the interactive processes between radioactive decay heat from the vitrified waste, infiltration of groundwater, swelling pressure generation and chemical evolution of the buffer material and porewater chemistry. In this program, numerical experiment system for the coupled THMC processes will be developed in order to predict the long-term performance of the near-field (engineered barrier and host rock) for various geological environments. The simulation code development has been started in FY 2001 and three development steps are planned, because (1)development will be continued for some years, (2)feasibility of numerical experiment have to be confirmed by using existing tools. This report presents the following items of the simulation code development for the coupled THMC processes. (1)First step of the simulation code development (2)Mass transport passways in compacted bentonite (3)Parallelization of the simulation code
Yamauchi, Toshihiko
Kankyo Kagakkai-Shi, 14(6), p.567 - 575, 2001/12
no abstracts in English
Sun, Y.*; Hakoda, Teruyuki; Chmielewski, A. G.*; Hashimoto, Shoji; Zimek, Z.*; Bulka, S.*; Ostapczuk, A.*; Nichipor, H.*
Radiation Physics and Chemistry, 62(4), p.353 - 360, 2001/10
Times Cited Count:22 Percentile:81.38(Chemistry, Physical)no abstracts in English
Nakatsuka, Toru; Oka, Yoshiaki*; Koshizuka, Seiichi*
Nuclear Technology, 134(3), p.221 - 230, 2001/06
Times Cited Count:17 Percentile:74.85(Nuclear Science & Technology)no abstracts in English
Suzuki, Satoru; Kawamura, Katsuyuki*
JNC TN8400 2001-005, 41 Pages, 2001/04
A correlation between molecular structure and a vibrational spectrum of interlayer water in Na-smectite was investigated by means of Molecular Dymamics (MDs) simulations. Detailed comparison of simulation results with IR spectroscopic observations for the water-smectite system indicated good agreement. Internal vibrational spectra of water were obtained by the Fourier transformation of velocty auto-correlation function of hydrogen atom. A stretching vibrational spectrum of interlayer water consisted of a broad band with a peak top around 3400cm and a sharp peak around 3650 to 3700cm
and a sharp peak around 3650 to 3700cm . The fomer broad band was assigned to O-H vibrations between water molecules as bulk water, while the latter band was attributed to O-H ones oriented to siloxane surface through hydrogen bonding. The hydrogen bond distance, determined as the shortest O-O distance by the radial distribution function (RDF), revealed that hydrogen bond distance between water and siloxane surface (O
. The fomer broad band was assigned to O-H vibrations between water molecules as bulk water, while the latter band was attributed to O-H ones oriented to siloxane surface through hydrogen bonding. The hydrogen bond distance, determined as the shortest O-O distance by the radial distribution function (RDF), revealed that hydrogen bond distance between water and siloxane surface (O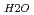 -O
-O
 3.0
3.0  -O
-O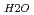 = ca. 2.8 AA ). These results suggested that interaction between water molecule and siloxane surface weaker than that between water molecules, although they were forced to be oriented.
= ca. 2.8 AA ). These results suggested that interaction between water molecule and siloxane surface weaker than that between water molecules, although they were forced to be oriented.
Fukumoto, Masahiro; Nishikawa, Yoshiaki*
JNC TN8400 2001-017, 355 Pages, 2001/03
The alkali hydrolysis experiments which seem to be important from the view point of the alteration mechanism using the following seven organic materials was performed as a part of the evaluation of the influence on the disposal of the organic materials contained in the TRU wastes. As a result of the alkali hydrolysis experiments (90 C and 91d), each organic materials became those of lower molecular weight. The degradation products were able to be detected in the solution. The organic materials seem to be degraded to the organic matters which were confirmed in this study in a long term of disposal. The degradation products were shown below. Therefore, the evaluation of the influence on the migration of radionuclides by degradation products becomes important in the future. (1)Cement additives of Naphthalenesulfonic acid and Ligninsulfonic acid (
C and 91d), each organic materials became those of lower molecular weight. The degradation products were able to be detected in the solution. The organic materials seem to be degraded to the organic matters which were confirmed in this study in a long term of disposal. The degradation products were shown below. Therefore, the evaluation of the influence on the migration of radionuclides by degradation products becomes important in the future. (1)Cement additives of Naphthalenesulfonic acid and Ligninsulfonic acid ( Naphthalenedisulfonic acid etc.) (2)Cement additives of polycarboxylic acid (
Naphthalenedisulfonic acid etc.) (2)Cement additives of polycarboxylic acid ( Oligomer of distal methoxypoly ethylene glycol.) (3)Ethylenediamine-N,N,N',N'-tetraacetic acid disodium salt (
Oligomer of distal methoxypoly ethylene glycol.) (3)Ethylenediamine-N,N,N',N'-tetraacetic acid disodium salt ( Acetic acid desorped and cyclized organic matters from EDTA.) (4)Tributyl phosphate (
Acetic acid desorped and cyclized organic matters from EDTA.) (4)Tributyl phosphate ( Dibutyl phthalate, n-butanol) (5)Poly vinyl acetate (
Dibutyl phthalate, n-butanol) (5)Poly vinyl acetate ( Acetic acid) (6)Nylon66 (
Acetic acid) (6)Nylon66 ( Adipic acid, Hexamethylenediamine) (7)Cured epoxy resin (
Adipic acid, Hexamethylenediamine) (7)Cured epoxy resin ( Glycerol poly glycidyl ether, Carboxylic acid)
Glycerol poly glycidyl ether, Carboxylic acid)
Makuuchi, Keizo; Haque, M. H.*; Ikeda, Kenichi*; Yoshii, Fumio; Kume, Tamikazu
Nihon Ratekkusu Arerugi Kenkyukai Kaishi, 4(1), p.7 - 10, 2001/01
no abstracts in English